A Smarter Model For Cancer Therapy
Our “3-S” cancer therapeutics are built to improve patient outcomes without adding complexity.
Watch our short animated explainer video to understand our technology, below.

Built for Patients.
Guided by Biology.
At Jabez, we identify and advance cancer therapeutics that are well-tolerated, affordable to produce, and seamlessly boost the efficacy of current treatment landscapes.
Following our strategic model, we see a path to real clinical progress that doesn’t require overhauling the healthcare system. It just takes some clever thinking, a deep understanding of cancer biology, and deliberate execution.

OUR APPROACH
"3-S" Cancer Therapeutics
Our “3‑S” framework is more than a slogan – it’s how every asset is vetted, licensed, and advanced.

Seamless
Easy-to-take: Orally available, outpatient-compatible, patient-friendly.

Scalable
Easy-to-make: Scalable chemistry, low‑cost manufacturing, global readiness.
Synergistic
Ready to integrate: Mechanistically synergistic with standard therapies.
JBZ-001:
Our Lead Compound
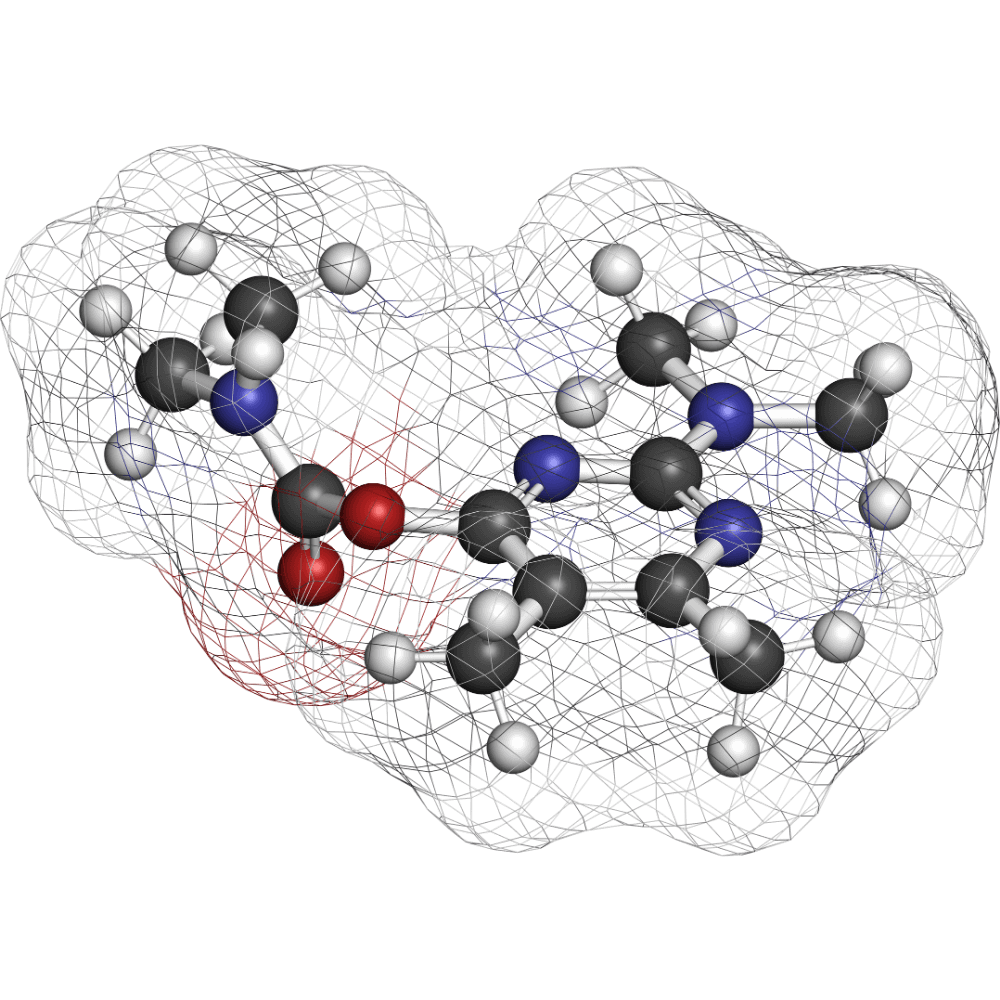
JBZ‑001 is an oral DHODH inhibitor in Phase 1 trials. It’s a proven 3‑S prototype with both single-agent promise and powerful synergy in combination.
It brilliantly exemplifies our 3-S model:
- Seamless: Well-tolerated, oral small molecule
- Scalable: Leverages Nobel Prize-winning, scalable chemistry
- Synergistic: Displays potent synergy with well-established treatment regimens.

CLINICAL DEVELOPMENT
Our Clinical Trials
Jabez Biosciences is proud to be a clinical-stage biotechnology company. We are currently enrolling patients in a Phase 1 clinical trial evaluating our lead candidate in advanced solid tumors and non-Hodgkin lymphoma.
Our clinical partners:



INVEST IN JABEZ
We’re raising investment to reach key clinical and commercial milestones.
We are now offering a pre-IPO investment opportunity, open to both accredited and non-accredited investors under a Regulation A+ offering.
Advance Our Lead Program
Establish safety, expand to synergistic indications (e.g., AML, MM), and demonstrate proof-of-concept for the Jabez model.

Demonstrate 3-S Clinical Impact
We aim to show that 3-S therapeutics can drive real-world outcomes – improving efficacy without added toxicity or logistical burden.

Expand Our Portfolio
We will identify and license additional assets that fit our 3-S filter to build a pipeline of smart, synergistic therapeutics.

Scale the Jabez Model
With each successful program, we expand our ability to systematically evaluate, acquire, and deploy high-impact therapies.
Latest
News & Events
- 08.18.25
- News
Jabez Biosciences, Inc. Opens Second Clinical Site in Phase 1 Oncology Clinical Study ofJBZ-001
- 05.09.25
- News
Jabez Biosciences, Inc. Announced Phase 1 Clinical Trial of JBZ-001 at AACR 2025
- 04.03.25
- News