At the heart of our scientific approach to cancer treatment is JBZ-001, our next-generation small molecule inhibitor of the enzyme dihydroorotate dehydrogenase (DHODH). DHODH plays an integral role in the production of nucleotides – vital building blocks required for cell division – and has been on scientists’ radar for several decades. Let’s dive into the scientific foundations of this technology and discover how we’re giving this classic therapeutic modality a modern makeover.
Jump to:
Understanding Cancer's Metabolic Vulnerabilities
At this very moment, most of the cells in our body are consuming nutrients such as sugars and amino acids, and transforming them into essential building blocks and energy to fuel cellular function.
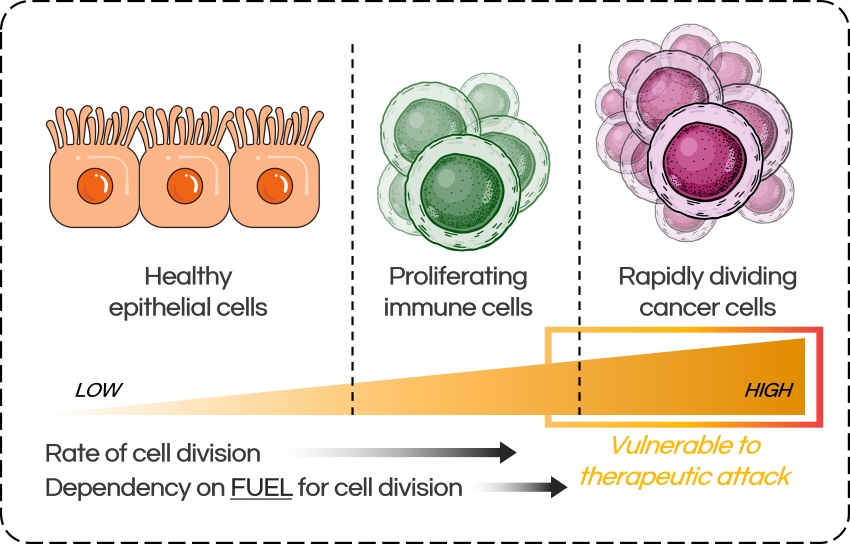
Cancer cells do the same, and since they divide very quickly, they require very high levels of these building blocks – much more than healthy cells. To meet the intense demand, they often rewire their metabolism, shifting the balance towards the production of vital building blocks.
In cancer biology, this is called “metabolic dysregulation,” and it’s a common feature of many types of cancer – especially aggressive, rapidly growing ones. While this shift in their metabolism allows cancer cells to grow quickly, it leaves them heavily reliant on certain biosynthetic pathways, and therefore, vulnerable to therapeutic attack.
DHODH Inhibitors: Exploiting Nucleotide Metabolism
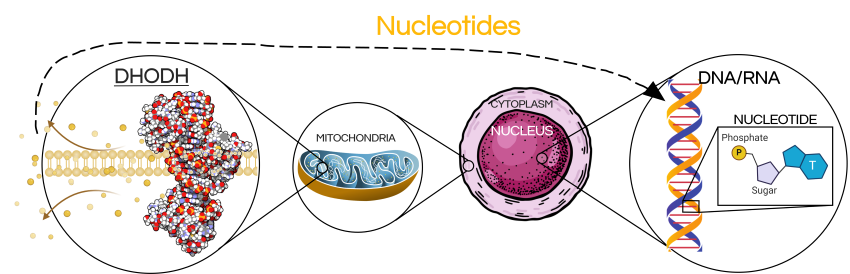
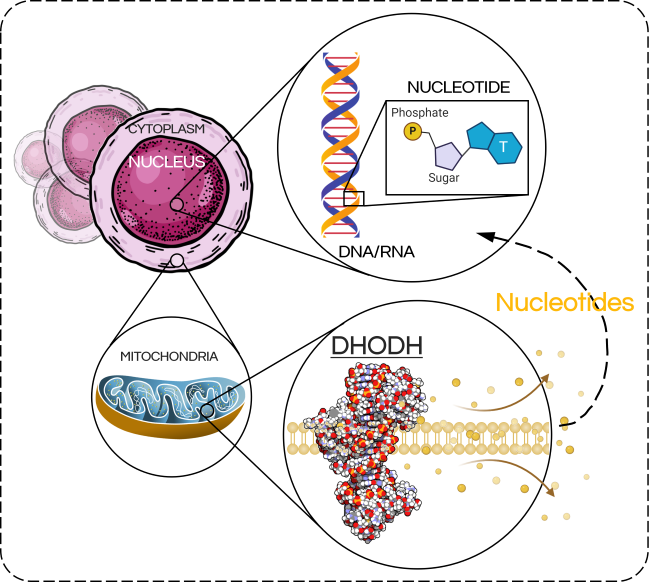
One of these essential building blocks is nucleotides, which the cell uses to build DNA and RNA – two extremely important macromolecules. Our cells require an enormous amount of nucleotides for proper function – for example, a normal human cell uses about 200,000 nucleotides EVERY SECOND during DNA replication – and, since they tend to divide more frequently, cancer cells require even more.
DHODH Inhibition: The Core Concepts
Dihydroorotate dehydrogenase (DHODH) is a mitochondrial enzyme that catalyzes a crucial step in the synthesis of pyrimidine nucleotides. Rapidly dividing cells (such as cancer cells), rely heavily on DHODH activity and it’s inhibition results in the swift depletion of nucleotides. This spells disaster for the cell, causing processes such as DNA replication and RNA transcription to grind to a halt, inducing massive amounts of cell stress and cell death.
DHODH In Human Disease: A Historical Perspective
DHODH has been on scientists’ radar as an important driver of cancer progression since the 1950s. Initial interest waned but in the intervening decades, a series of scientific breakthroughs has rekindled scientists’ interest in this classic therapeutic target. Let’s investigate the history of this field and explore the technological and scientific breakthroughs that brought us here.
JBZ-001
Our Next-Generation DHODH Inhibitor
Developed by leading scientists at The Ohio State University James Cancer Hospital and Solove Research Institute, our lead compound JBZ-001 is the culmination of decades of scientific advancements and represents a significant leap forward in this therapeutic class.
Superior Potency
Optimized Pharmacokinetics
Enhanced Safety Profile
Efficient Manufacturing
Learn more, below.
Superior Potency
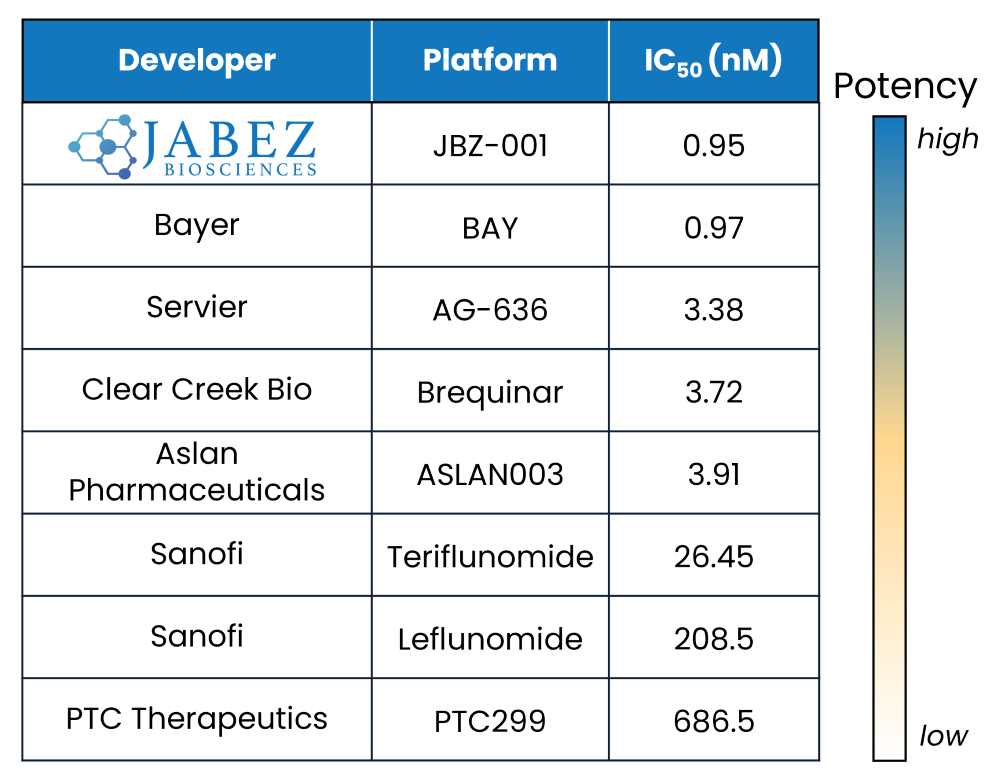
In our recent cell-free DHODH inhibition assay, JBZ-001 demonstrated unparalleled potency with an IC50 of just 0.95 nM, outperforming all prominent DHODH inhibitors currently available. This exceptionally low IC50 value highlights JBZ-001’s superior ability to inhibit DHODH, underscoring its position as a potentially superior therapeutic option.
Optimized Pharmacokinetics
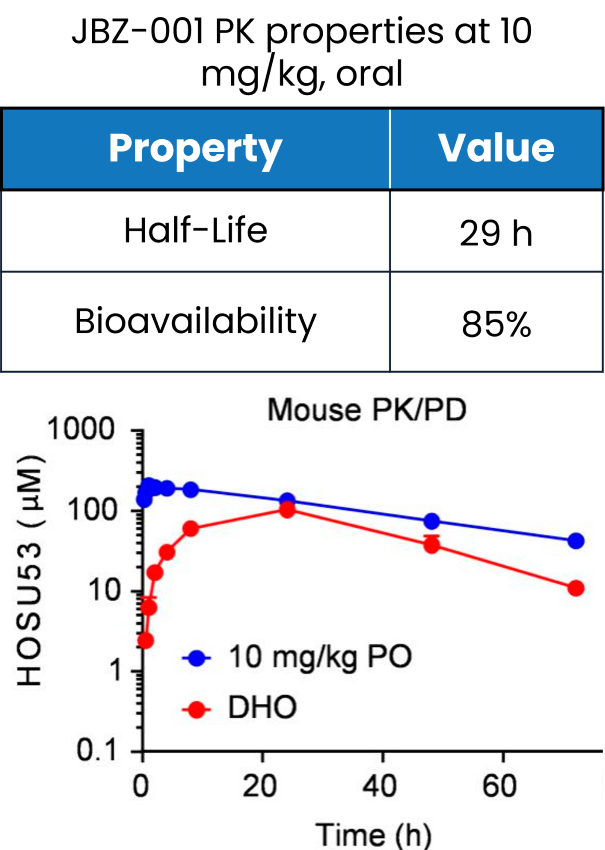
Our pharmacokinetic study in mice revealed JBZ-001’s impressive oral bioavailability at 85%, alongside a prolonged terminal half-life of 29 hours (blue line). These findings confirm JBZ-001’s efficient absorption and sustained activity, further supporting its therapeutic potential.
Wider Therapeutic Window
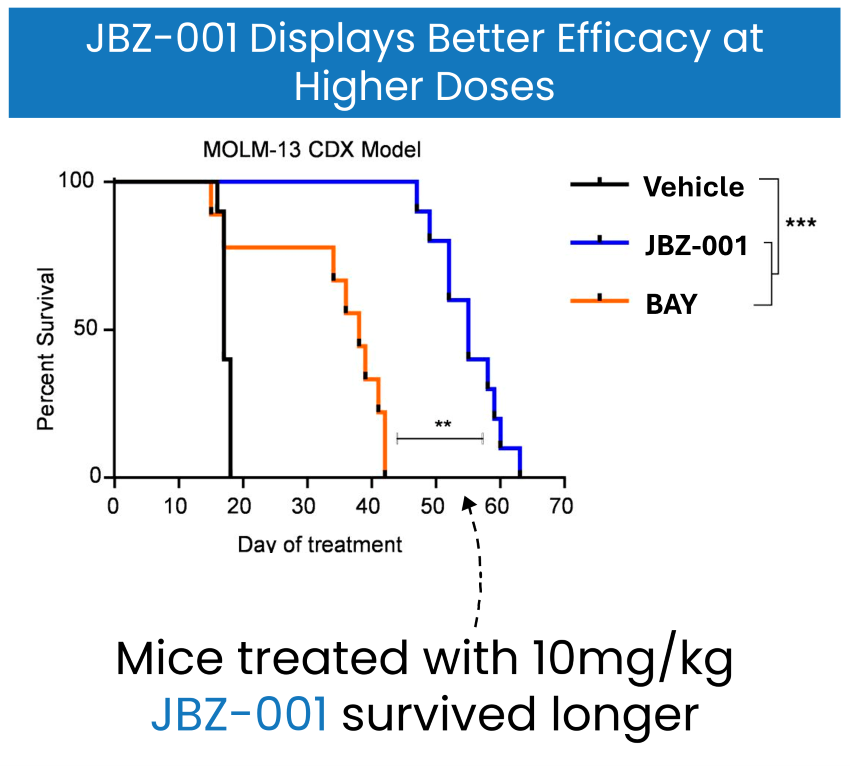
JBZ-001 exhibits a superior safety profile with a wider therapeutic window, tolerable at doses of 10 to 30 mg/kg. In contrast, the next most potent DHODH inhibitor BAY (orange line) has a maximal tolerable dose of around 4 mg/kg and is not tolerable at 10 mg/kg. At this higher dose, JBZ-001 (blue line) remains safe and shows significantly better performance in mouse models.
Efficient Manufacturing
The synthesis of JBZ-001 is highly efficient, utilizing the Suzuki reaction, a preferred method for forming carbon-carbon bonds in pharmaceutical manufacturing. This reaction employs stable, nontoxic, and cost-effective substrates, operating under mild conditions, which makes it environmentally friendly and scalable for industrial production. The straightforward process enables the production of therapeutically relevant quantities of JBZ-001, enhancing its commercial viability.
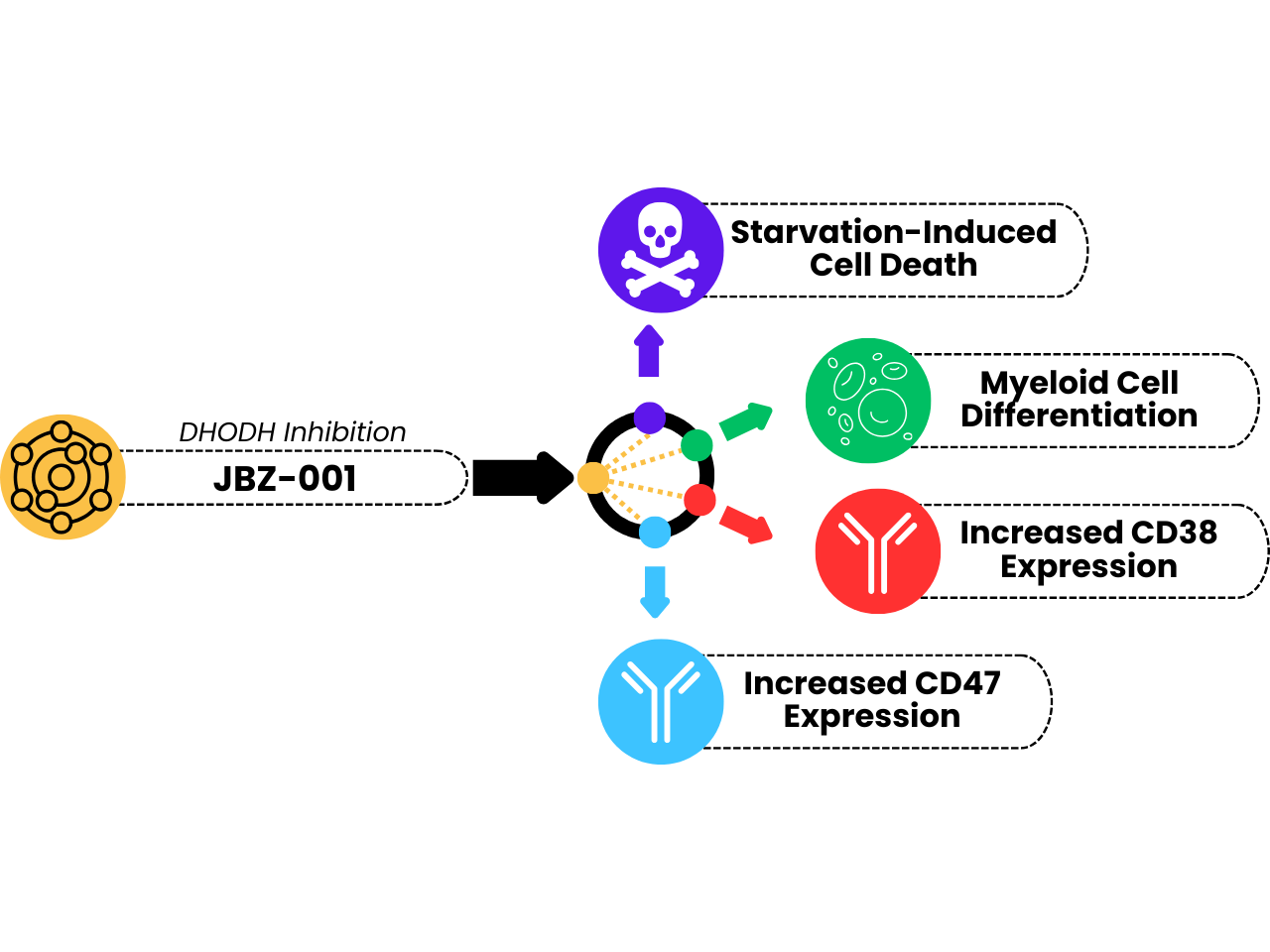
Diverse Therapeutic Effects
On top of the expected nucleotide depletion-induced cell stress, JBZ-001 displays multiple mechanisms of action, broadening its potential clinical applications, including:
- The differentiation of myeloid cells, which is significant for treating myeloproliferative disorders such as acute myeloid leukemia (AML).
- Enhances the expression of CD38, boosting the efficacy of anti-CD38 therapeutic antibodies, a key strategy in treating multiple myeloma.
- Increases CD47 expression, augmenting the effectiveness of anti-CD47 “Don’t Eat Me” checkpoint blockades.
Understanding DHODH Inhibition
Therapeutic Mechanisms of Action of JBZ-001
On top of the expected nucleotide depletion-induced cell stress, our DHODH inhibitor, JBZ-001, displays multiple mechanisms of action, opening up the possibilities to be used in combination with currently approved therapies.
Explore JBZ-001’s Therapeutic Potential, below:
Our Pipeline
We are actively pursuing clinical investigations in various solid tumor indications and non-Hodgkin lymphoma (NHL), reflecting our commitment to addressing a broad spectrum of oncological needs. Explore our pipeline to see our current and future plans.